RNA regulation in childhood cancer
Gene expression is a highly dynamic process. To regulate RNA synthesis and degradation, cells have a variety of tools for regulating gene expression.
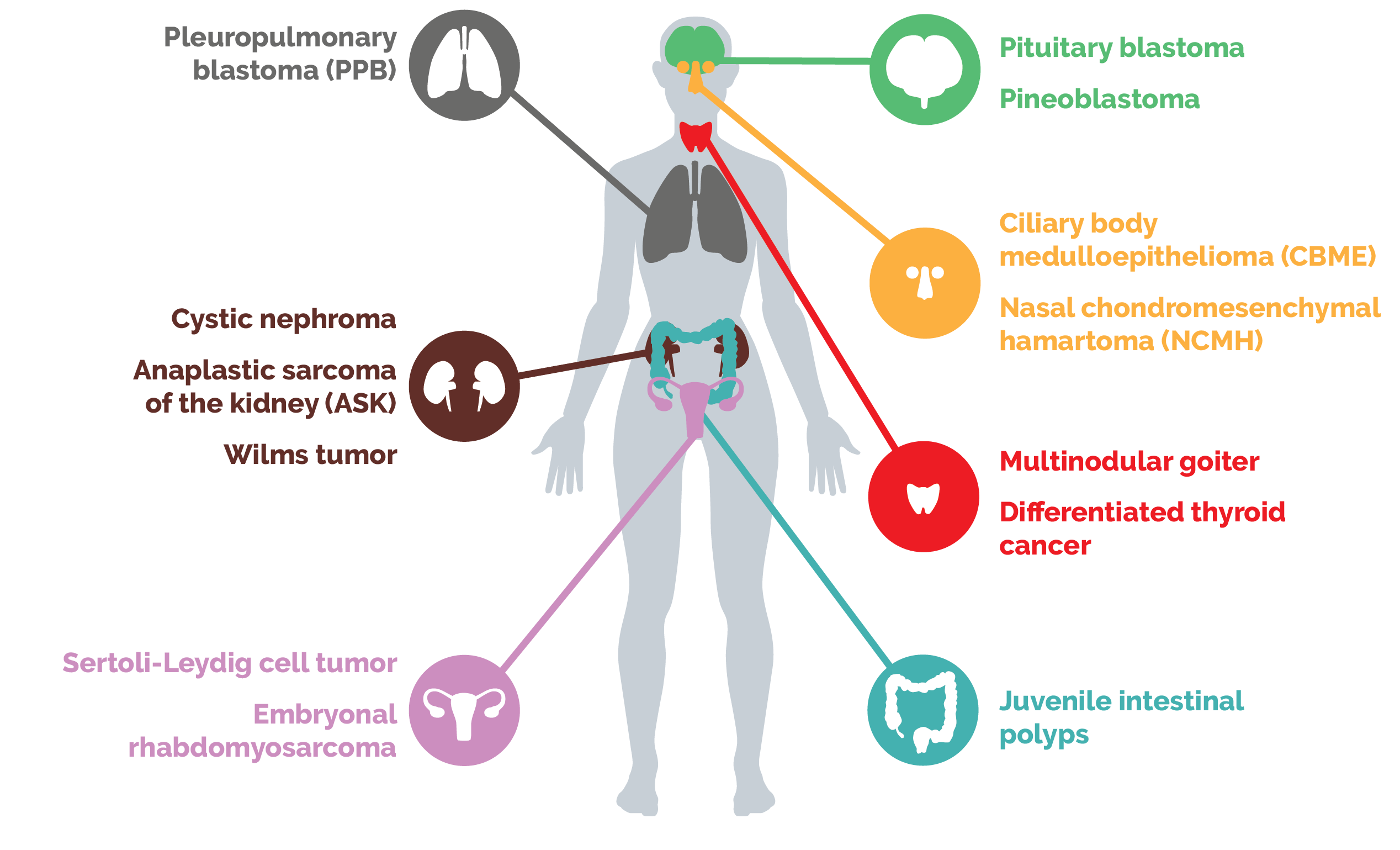
However, many childhood cancers are driven by loss of these regulatory tools. For example, loss of one of these tools, known as microRNAs, can lead to a variety of rare childhood tumors. Many other cancers bear mutations in transcription factors, the proteins that give a cell its identity by determining which genes are expressed. Yet other cancers are driven by mutations in another regulatory tool, the chromatin remodeling complexes.
We study how dysregulation of RNA synthesis and degradation drives childhood cancers with the ultimate goal of identifying new therapeutic vulnerabilities to exploit in treating them.
Scroll down to learn more.